Building the leading RNA medicines company
Our RNA medicines platform, PRISMTM, combines multiple modalities, chemistry innovation and deep insights in human genetics to deliver scientific breakthroughs that treat both rare and prevalent disorders.
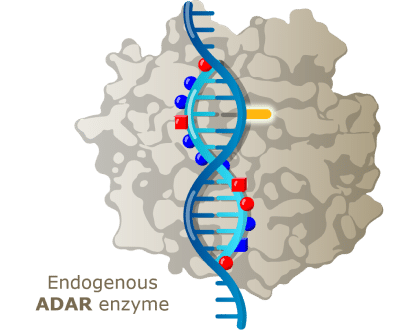
What is an “RNA medicine”?
An RNA medicine is a short strand of synthetic ribonucleic acid (RNA) or deoxyribonucleic acid (DNA) connected by chemical linkages (also referred to as an oligonucleotide). Wave’s oligonucleotides are designed to engage messenger RNA (mRNA) strands inside human cells to repair, restore, or reduce proteins to treat diseases.
RNA medicines provide advantages over other types of medicines, including the ability to:
- Avoid risk of permanent off-target genetic changes associated with DNA-targeted approaches
- Modulate the enormous transcriptional diversity of cells
- Address diseases that have historically been difficult to treat with small molecules or biologics
- Access a variety of tissue types or cell types throughout the body
- Modulate the frequency of dosing to ensure broad distribution within tissues over time
- Leverage well-established industry manufacturing processes and regulatory, access, and reimbursement pathways

Unlocking
what’s possible
Our scientific approach is based on the recognition that the biological machinery necessary to address human diseases already exists in our cells and can be harnessed for therapeutic purposes with the right tools.
We have built the most versatile platform to develop RNA medicines in the industry, with multiple means of repairing, restoring, or reducing proteins depending on the unique biology of a given disease target and treatment goals. This core capability sets us apart from other companies and enables us to pursue the optimal treatment approach for each disease.
Our modalities
Pioneering Best-in-Class Chemistry
Since our founding, we have deftly challenged convention related to oligonucleotide chemistry. This led us to discover and develop several proprietary, novel innovations that enable us to optimize the pharmacological properties of our molecules, thereby enhancing potency, distribution, and durability in the body.
Due to these chemistry modifications, we can avoid the use of complex delivery vehicles, such as lipid nanoparticles and viruses. Instead, our candidates for liver diseases use clinically proven N-acetylgalactosamine (GalNAc)-conjugates or are freely taken up in a variety of other cell types, including neurons in the central nervous system, muscle cells, and more.
Wave maintains strong and broad intellectual property on our novel chemistry modifications, including stereochemistry, phosphoryl guanidine-containing backbone linkages, and others.